Double redox aqueous capacitor with high energy output
- Post by: nfp
- 01/03/2023
- Comments off
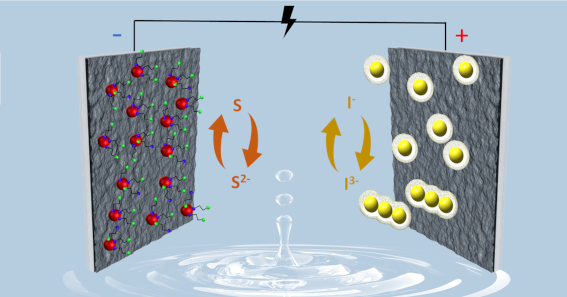
The paper puts forward the concept of double redox electrochemical capacitor operating in an aqueous electrolyte. It combines the redox activity of sulphur from Bi2S3 nanocrystals combined with the reactions of iodides which take place at opposite electrode. This combination effectively boosts the capacitance (275 F g-1), and the energy density of the device thanks to relatively high cell voltage of 1.5 V. The device retains 73% of its capacitance at 10 A g-1 of discharge current. The different states of the redox species ensure their operation at separate electrodes in an immiscible manner without a shuttling effect. The specific interactions of the redox active species with carbon electrodes are supported by operando Raman spectroscopy.
Full article here.
NFP Research Area: Nanomaterials for energy, environment and safety: Nanomaterials For Solar Energy Harvesting
